Sustainable Reaction Engineering Laboratory
In the Sustainable Reaction Engineering laboratory, research focuses on heterogeneous catalysis and reactor design, with the goal of developing highly efficient processes that use renewable energy to convert greenhouse gases into sustainable synthetic fuels. This includes converting carbon dioxide that would otherwise pollute the environment into a sustainable and affordable fuel would minimize greenhouse gas emissions and reduce our dependence on fossil fuels.
Research Spotlight
Catalyzing a Lower-Carbon World
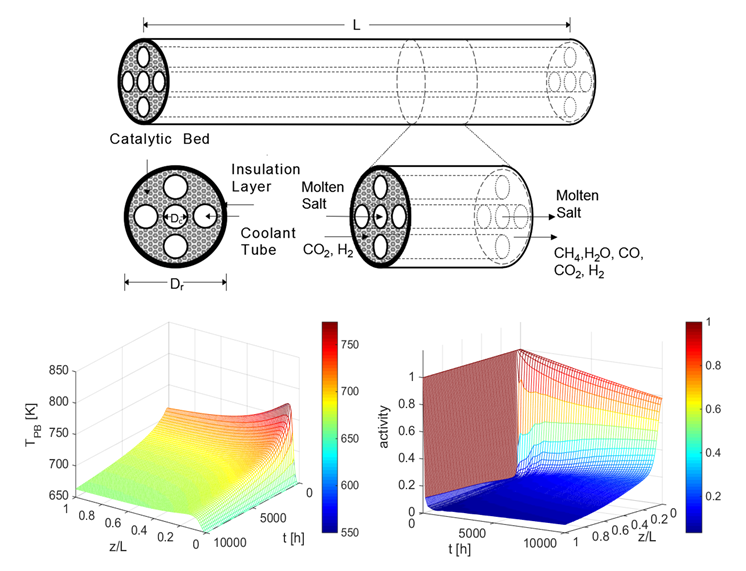
The more carbon dioxide (CO2) we pump into the atmosphere, the hotter the planet will get. That’s why researchers around the world are looking for ways to transform those emissions into useful resources. One promising approach is to convert them into methane fuel, using a hydrogenation process called the Sabatier reaction.
In theory, it’s simple: just add hydrogen gas to CO2 in the presence of a catalyst. However, there are technical challenges. Because the process generates a lot of heat, the reactor must be constantly cooled. It’s also crucial to limit the creation of carbon deposits that can deactivate the catalyst.
WISE researcher David Simakov is addressing those issues. He and his chemical engineering team have developed a mathematical model of an actively cooled Sabatier reactor...
Lab Gallery
Contact
(519) 888-4567 x37048 Email |
Page Options

